Pioneering Sensing Technology Since 20101
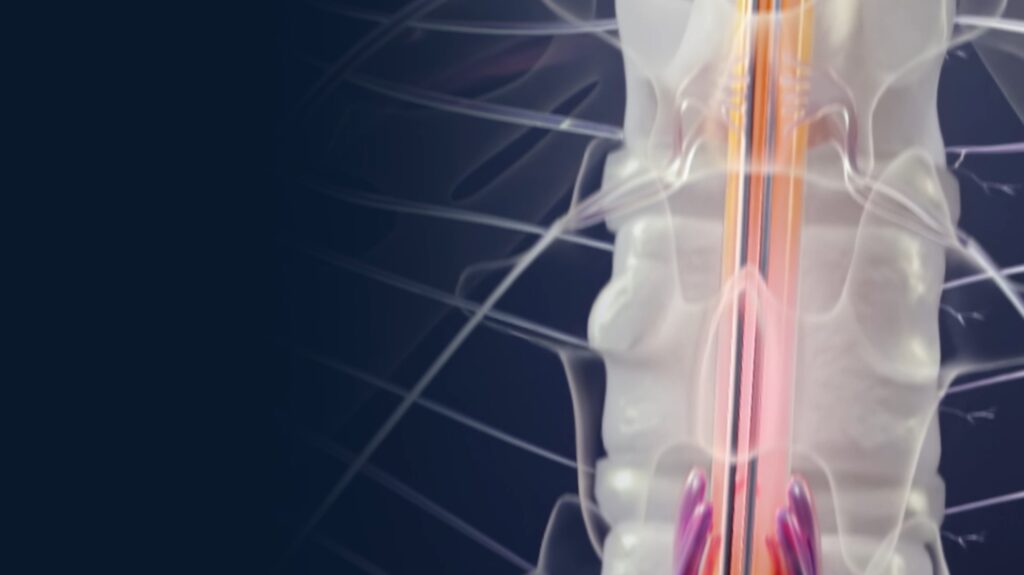
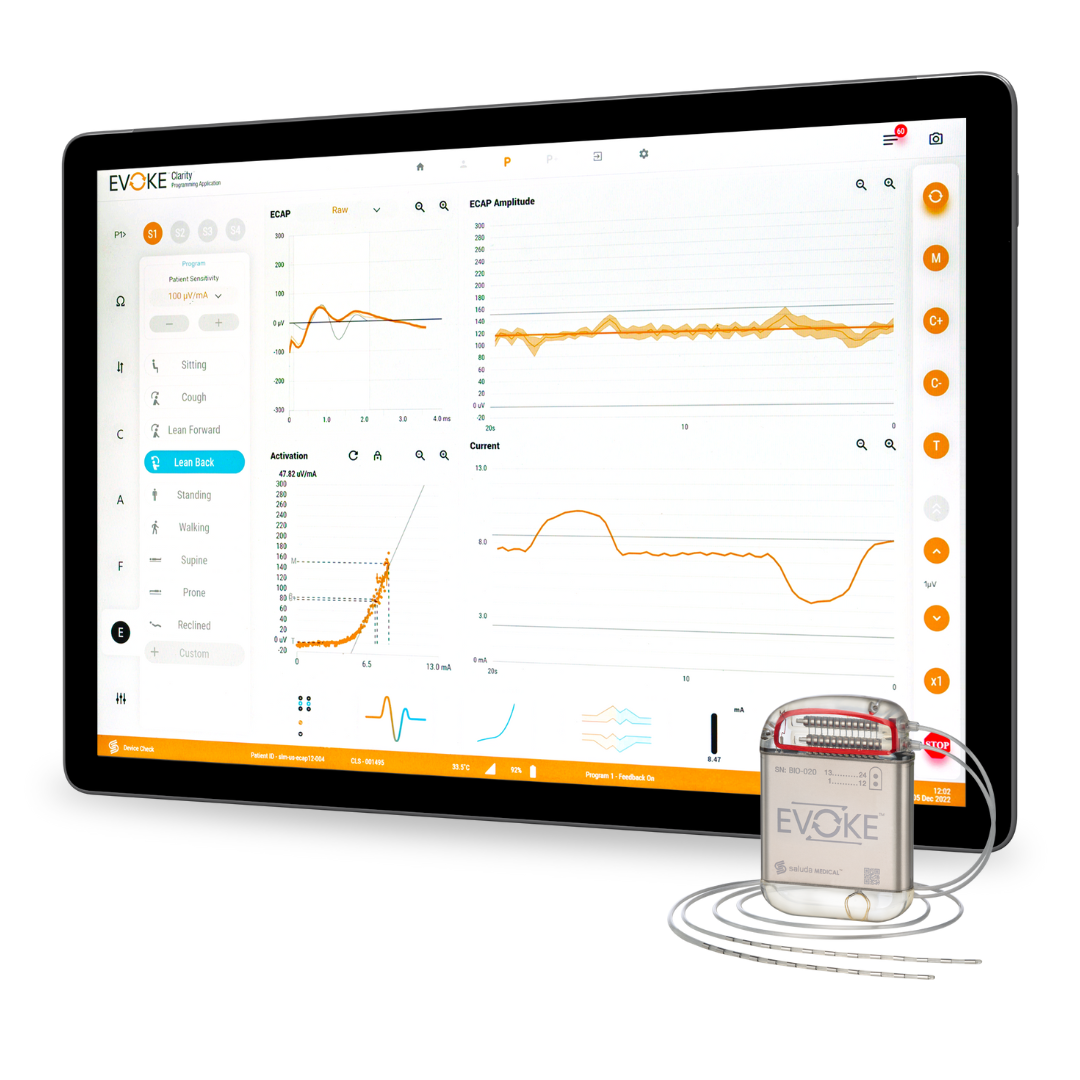
With over a decade of research, development, and clinical experience, we’ve built the first SCS system that directly records the spinal cord’s physiologic response to stimulation using Evoked Compound Action Potentials (ECAPs).
First to SENSE
In 2011, we sensed and recorded the spinal cord’s response to stimulation (an ECAP) for the first time.

Proven by SCIENCE
The Evoke® System was built on 15+ years of research and clinical experience.

Leading the FUTURE
Our sensing technology is leading to a new generation of neuromodulation therapies.

Dr. Jason Pope, MD
Evoke® System for Providers

Jennifer R. | Evoke™ Therapy Patient